 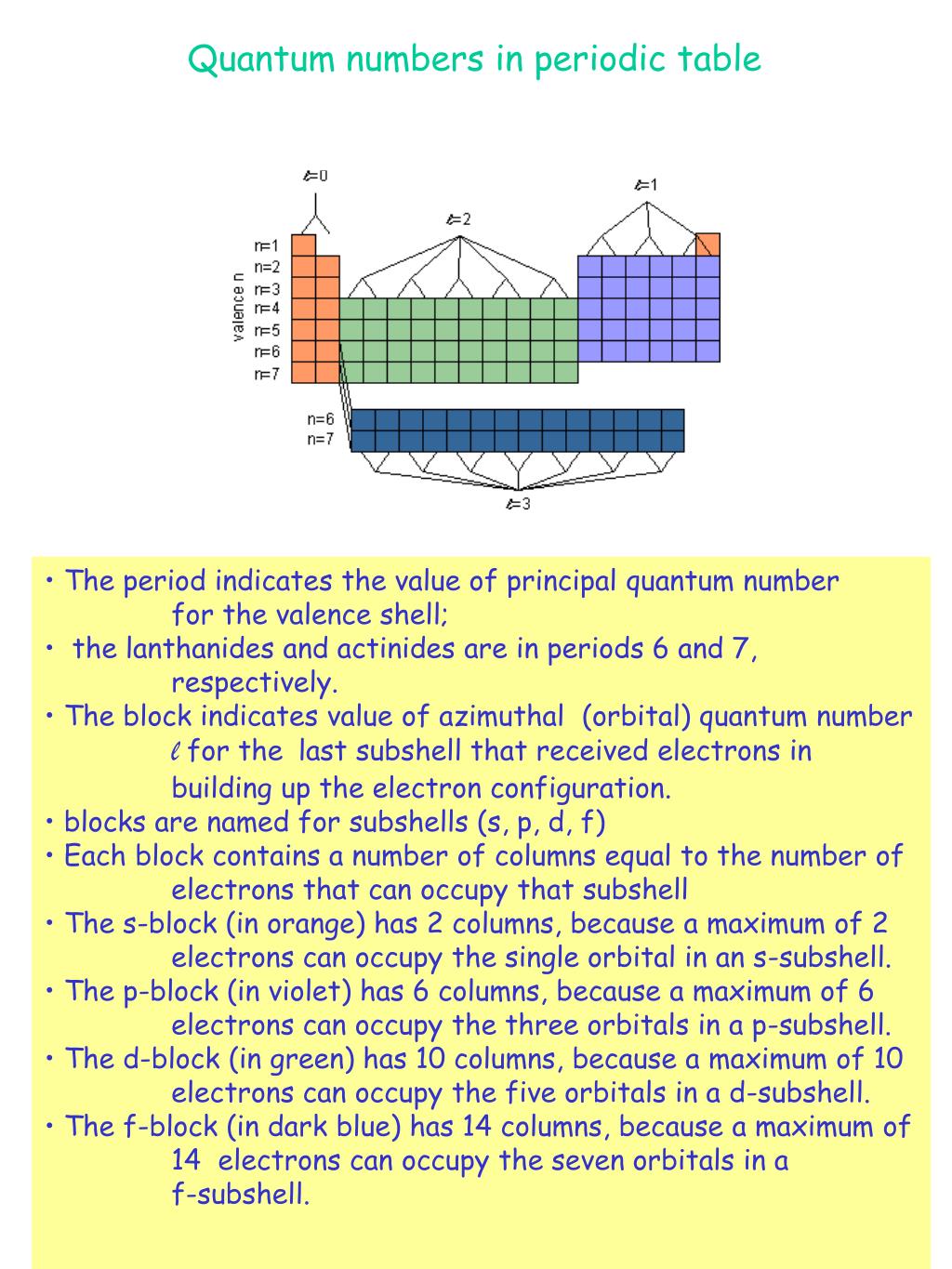
These values are denoted as letters s, p, d, and f. For l, the value is a range from 0 to ( n – 1). The angular momentum ( l) and magnetic ( m) numbers are dependent on the principal number. The principal quantum number ( n) determines the size of the orbital and its values can range from 1, 2, 3 and so forth. The values of the three quantum numbers are given in integers. Magnetic ( m) – The orientation of orbital in space and the number of orbitals.Angular Momentum ( l) – The shape of the orbital. :max_bytes(150000):strip_icc()/AlkaliMetals-56a12cd73df78cf772682671.png)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
